The manipulation of materials has characterized the great moments of human evolution throughout its history. The ages of stone, bronze and iron owe their name to the materials that man learned to use at that time and that managed to develop that era. The great technological advances experienced in the twentieth century are related in large part to the use of silicon. The 21st century has brought with it the development of a wide group of materials that have attracted attention thanks to the diverse physical, chemical and morphological properties they present and the wide range of applications they have. These are the six most outstanding.
Synthetic Diamonds
Synthetic Diamonds are one of the most sought after and desirable super materials in the world today. It is a very diverse material that has found use in many different industries. This is because the material is very efficient in tough environments, thanks to its abrasive, mechanical, optical, thermal and electro-chemical properties. Its industry use has been observed but not limited in healthcare, auto production, recycling and more. For more information about the material and its use check the ebook on Element 6.
Carbon nanostructures
They are perhaps the most famous in the last decade. Among them are the carbon nanotubes, fullerenes and graphene, which are part of the same family since they are all different allotropic forms of carbon. They are characterized by being extremely hard, resistant and flexible and have been used in rechargeable batteries, auto parts, sporting goods and water filters. Fullerenes support high pressures and have very good ability to combine with other elements, so they have been used for controlled release of drugs. Graphene is the thinnest material in the world and has much elasticity, is stronger than steel, better electrical conductor than copper, transparent and antimicrobial. Its applications range from curved cell screens, fast charge batteries, fuel cell shields and more.
Two-dimensional semiconductor materials
A disadvantage of graphene is that it is not a semiconductor, which limits some of its applications. In the search for semiconductors, very interesting materials have been obtained, such as phosphorus, a material composed of layers of a phosphorus atom organized in a hexagonal structure. Although it was synthesized only in 2014, its applications in electronics are very encouraging. In this same family are silicon and antimony, materials formed by sheets of silicon and antimony atoms, respectively. The first has been used to build batteries with greater efficiency and lifespan. Antimony has proven to be highly stable in different media and is projected for applications in optoelectronic technologies such as new light-emitting diodes (LEDs).
Nanoporous materials
Applications such as purification, separation and storage of gases, adsorbents and molecular filters have attracted attention in recent times due to the contemporary environmental challenge. And porous materials play a big role in this. Among them, metal-organic networks have excelled. Their unique characteristics of high porosity, high internal surface area, flexibility and ability to incorporate specific functionalities make them special and have increased their research. Another example of striking nanoporous material is the upsalite, a magnesium carbonate obtained in 2011 at the University of Uppsala (Sweden), which has surprised with its super absorbent properties.
Topological materials
Topology is a branch of mathematics that studies the properties that do not change in geometric bodies under deformations. Applying it to the study of materials allowed the discovery of new systems with exotic properties. Among them are topological insulators (materials that are electrical insulators inside and conductors on the surface), topological superconductors (materials that can conduct electricity without losing energy in the form of heat thanks to the action of Majorana fermions, particles that are their own antiparticle) and topological semimetals (they have the same electronic properties of graphene but in 3D). Its applications focus on the manufacture of devices that, like the quantum computer, will allow processing and solving problems millions of times faster than current devices.
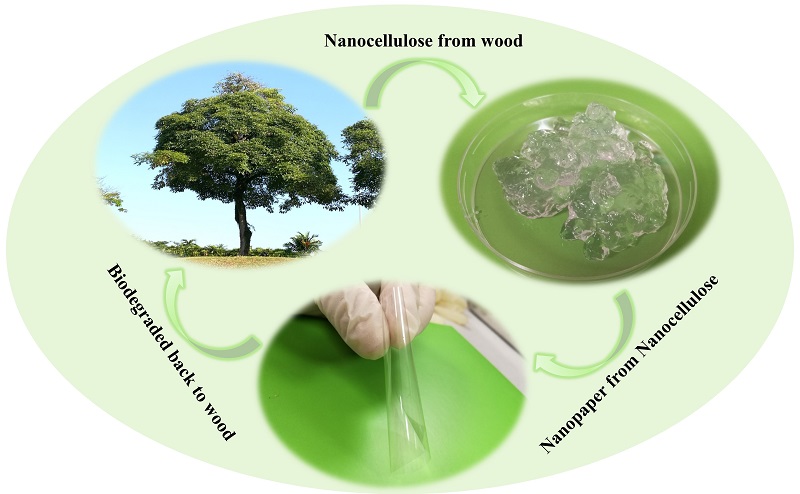
Nanocellulose
An immediate environmental challenge is the replacement of plastic, a polymer that represents a serious problem due to its difficulty in decomposing. Among the best candidates is nanocellulose, a material that is obtained based on cellulose that has been decomposed, purified and reconstructed in nanometric dimensions. Cellulose is the most abundant biopolymer in nature and is one of the structural components of plants, although it is also found in algae and some marine animals. Once processed it is possible to obtain nanocellulose, which has surprised by being a material resistant to high temperatures, flexible, easy to modify chemically, transparent and good conductor of electricity. But its biggest advantage is that it does not generate polluting waste, which makes it a totally ecological nanomaterial with zero environmental impact. Creams for the skin, new textiles, paper and improved cements are other examples of products where nanocellulose is being used with satisfactory results.
There is no doubt that the future is increasingly within our reach and it will be the new materials that will help us to build a new era.